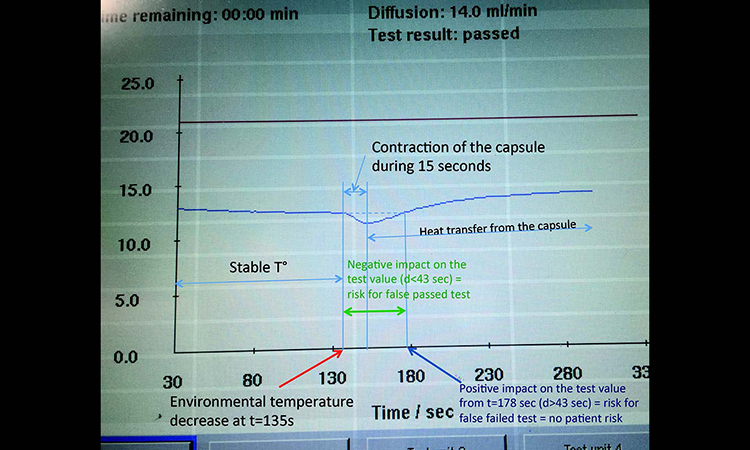
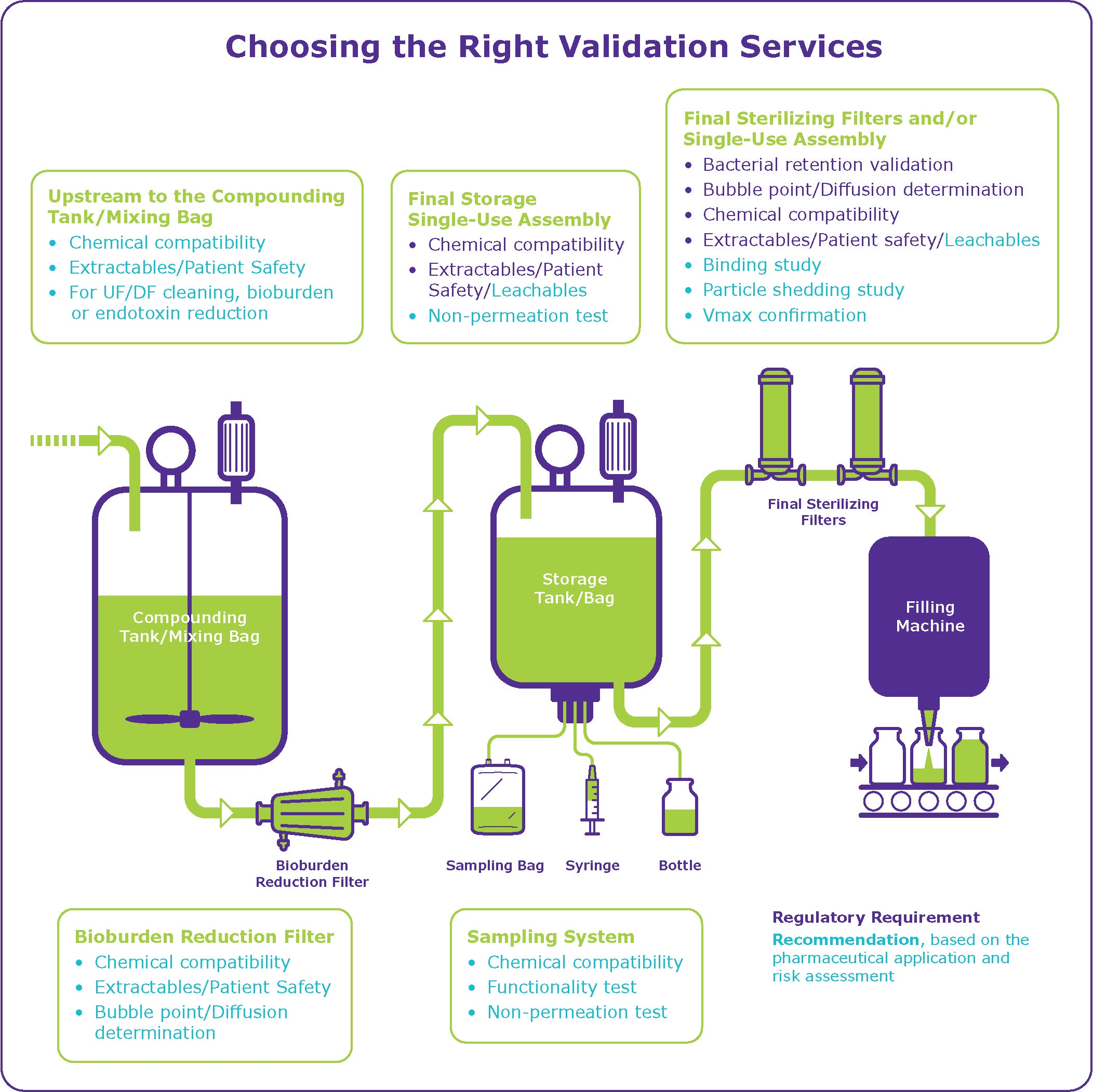
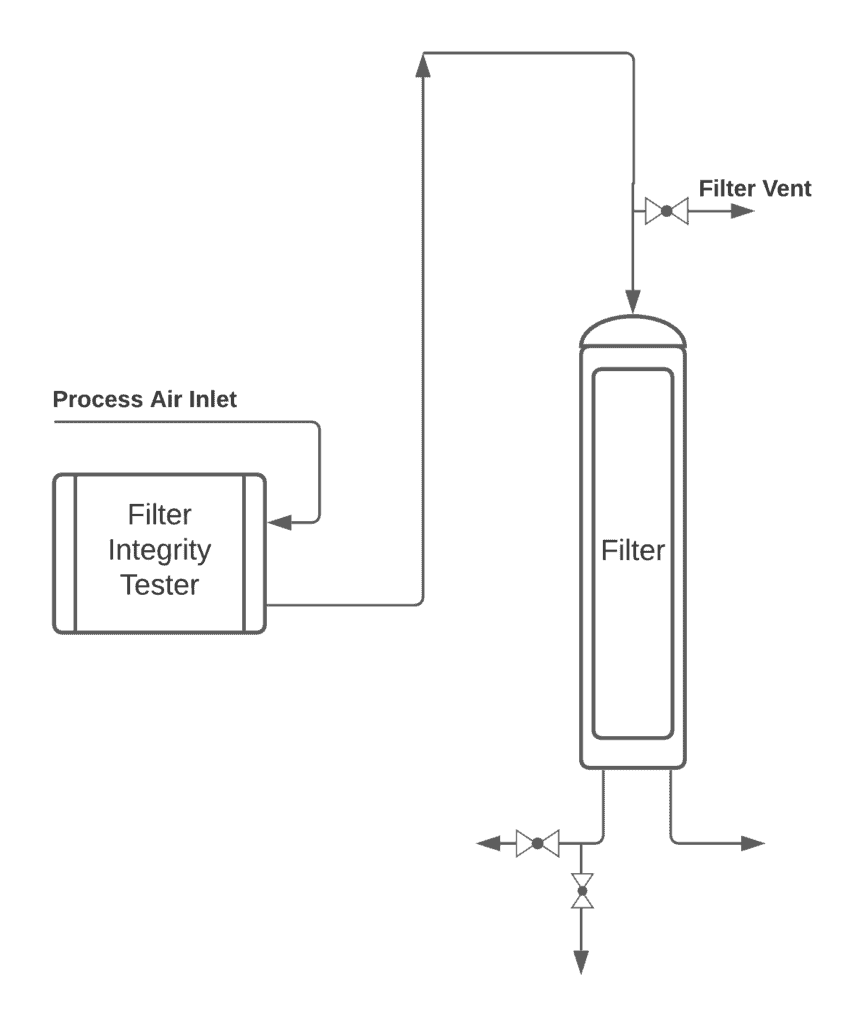
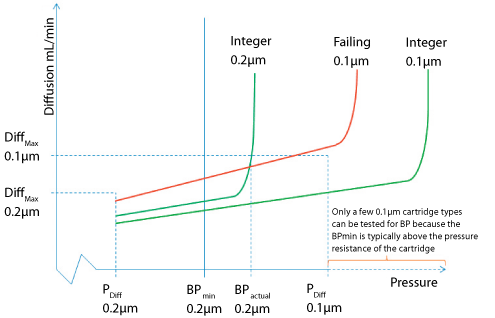
Pre-use/post-sterilization Integrity Testing of Sterilizing Grade Filter: The Need for Risk Assessment | American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology

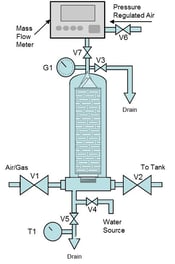
China Lab Filter Integrity Tester Include Water Intrusion Method Bubble Point Test - China Filter Integrity Tester, Water Intrusion Method