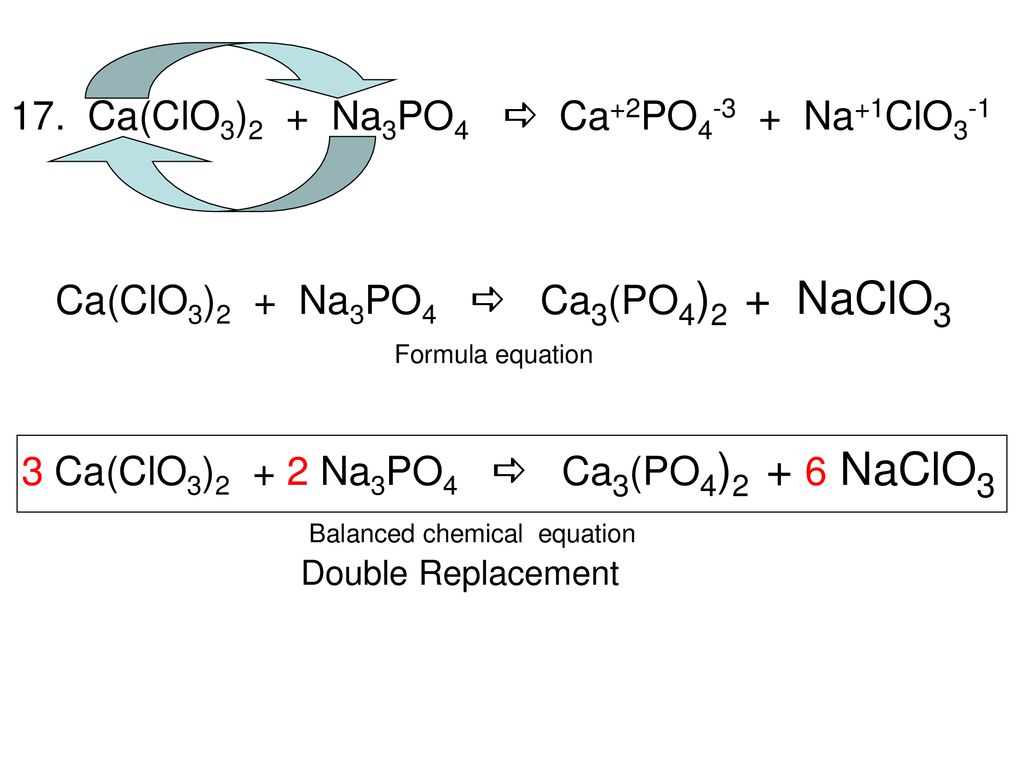
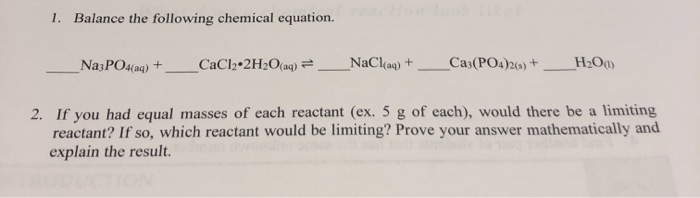Solved] If you have9 moles of CaCl2and all the Na3PO4I needed, how many moles of NaCl could you produce? | Course Hero

Chemical Equations Chapter 11 Notes. Balanced Equation 2 NaCl + F 2 2 NaF + Cl 2 ReactantsProducts Yields is the “equals” Coefficient used to balance. - ppt download

OneClass: I have started this problem but now I am stuck. Please show all work on how the answer is ...